SCANRDI®
Bring your Sterility Testing to the Next Level
SCANRDI® revolutionized rapid microbial detection when it was first introduced and still today its speed and sensitivity remain unrivaled.
Its unique technology delivers actionable sterility results in 4 hours or less, so you know you can continue with production or if you need to take action immediately.

- Features
- Tests
- Downloads
- Services
- Videos
When you Need to Know Now
SCANRDI® enables same-shift detection of microbial contaminants in filterable drug products. The US Food and Drug Administration (FDA), European Medicines Agency (EMA), and other regulatory bodies support the use of validated Rapid Microbiological Methods (RMM) for sterile product release - especially in the case of shelf-life products, where a rapid result is more suitable than a compendial 14-day test.
Ultra-rapid
- Same-shift results
- Release your product faster
Accurate
- Tested & proven technology
- Make decisions with confidence
Compliant
- Full validation support: URS completion guide, IOQ guide, primary validation guide, suitability guide and Drug Master File (DMF)
- 21 CFR part 11 enabled software
Proven Technology
- Long history of use for final product sterility, in-process bioburden control & water testing
- Down to 1 microbe/filtrable volume
- >200 compatible products
Broad Specificity
- Universal fluorescent labeling
- Bacteria, yeast, and mold detection
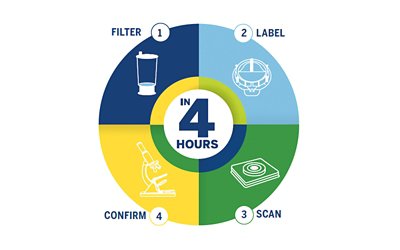




- Filter
- Label
- Scan
- Confirm
Improve productivity and compliance
Increase Your Market Competitiveness
- Safely release your short shelf-life & generic products faster
- Optimize your distribution logistics with just-in-time inventory management of stock and choose your most affordable shipping option
Control Your Manufacturing Process
- Rapid in-process, bioburden and sterility results give you confidence that your production is operating properly
- Facilitate your investigation process to quickly return to normal operations
- Eliminate lab bottlenecks
Be Confident in Your Compliance
- Ensure your electronic compliance with 21 CFR part 11 enabled software, barcode scanning traceability, several approval levels and a full audit trail
- Feel reassured in your validation process with support from bioMérieux including URS completion guide, IOQ guide, primary validation guide, suitability guide and Drug Master File (DMF)
More than 200 Compatible Products*
- Antibiotics
- Isotonic solutions
- Chemotherapy drugs
- Hormones
- Anti-inflammatories
- Anti-coagulants
- Analgesics
- Diuretics
- Vitamins
* non exhaustive list
SCANRDI® is a rapid, proprietary microbiological solution combining universal cell labeling and solid-phase cytometry with the ability to detect bacteria, yeasts, molds – both in vegetative and sporulated forms - and yeasts. Test applications include:
- Raw material testing
- Water testing
- In-process product testing
- Chromatography columns health check
- Investigations
- Product release
As it’s a non-based growth method, stressed and fastidious organisms including those in VBNC state (Viable But Non Culturable) can be detected as well, making SCANRDI® a powerful tool to monitor and ensure the rapid detection of microorganisms in pharmaceutical products.
Downloads
Brochure - SCANRDI® - When you Need to Know Now Bring your Sterility Testing to the Next Level
Download
- Filename
- BROCHURE_SCANRDI_A4_05_23_9324097_Pages.pdf
- Size
- 595 KB
- Format
- application/pdf
Services: Feasibility, Validation, and Instrument Support
With bioMérieux services, your productivity and product quality is our priority. Our mission is to provide the pharmaceutical industry with rapid, accurate, and cost-effective solutions to ensure smooth implementation and maximizing your return on investment.
Feasibility Studies
- Quickly confirm the compatibility of your product and method
Validation Services
- Expedite implementation of your investment by reducing validation time and expense
Instrument Service Plans and Support
- Maximize the uptime of your instruments and ensure continuous operations
Smart Remote Support Solutions
- VILINK® ensures the continuity of your operations

