PARTE 6 - Explicação da detecção de deterioração da cerveja por PCR
Explicação da detecção de deterioração da cerveja por PCR
 Todos os microrganismos deteriorantes de cerveja possuem DNA (ácido desoxirribonucleico) que contém as instruções genéticas para o desenvolvimento e função dos organismos vivos. Enquanto nos animais, plantas e leveduras o DNA está contido no núcleo da célula, nas bactérias o DNA está presente no seu citoplasma (o material interno à célula, delimitado pela membrana celular).
Todos os microrganismos deteriorantes de cerveja possuem DNA (ácido desoxirribonucleico) que contém as instruções genéticas para o desenvolvimento e função dos organismos vivos. Enquanto nos animais, plantas e leveduras o DNA está contido no núcleo da célula, nas bactérias o DNA está presente no seu citoplasma (o material interno à célula, delimitado pela membrana celular).
A reação em cadeia da polimerase (PCR) é um teste simples e elegante inventado em 1983 pelo bioquímico americano Kary Mullis. Como descrito por ele na sua publicação (1990), ela "permite escolher o pedaço de DNA que te interessa e ter a quantidade que quiser". São necessárias apenas quantidades ínfimas de DNA para o PCR gerar cópias suficientes para serem analisadas utilizando métodos laboratoriais convencionais. Por esta razão, o PCR é um teste sensível.
Hoje em dia é um método amplamente utilizado em biologia molecular. Embora há 5 anos ainda fosse considerada uma tecnologia que só poderia ser encontrada em grandes laboratórios centrais e institutos de pesquisa devido ao investimento necessário, tanto em equipamento como em condições laboratoriais específicas. Mais recentemente, houve significativa simplificação de protocolos e equipamentos de PCR (termocicladores) que permite uma "democratização" da tecnologia PCR para sua utilização local como uma verdadeira ferramenta de produção, na cervejaria e ao longo de toda a cadeia de produção alimentar.
Em que consiste o DNA?
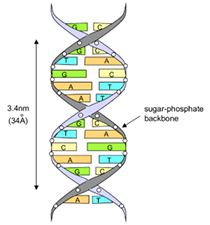 No DNA, a informação é armazenada como um código composto por quatro bases químicas: adenina (A), guanina (G), citosina (C), e timina (T). As bases de DNA emparelham-se entre si, A com T e C com G, para formar unidades chamadas pares de bases. Cada base está também ligada a uma molécula de açúcar e a uma molécula de fosfato. Juntas, uma base, açúcar e fosfato são chamadas de nucleotídeos. Os nucleotídeos estão dispostos em dois longos filamentos que formam uma espiral chamada dupla hélice.
No DNA, a informação é armazenada como um código composto por quatro bases químicas: adenina (A), guanina (G), citosina (C), e timina (T). As bases de DNA emparelham-se entre si, A com T e C com G, para formar unidades chamadas pares de bases. Cada base está também ligada a uma molécula de açúcar e a uma molécula de fosfato. Juntas, uma base, açúcar e fosfato são chamadas de nucleotídeos. Os nucleotídeos estão dispostos em dois longos filamentos que formam uma espiral chamada dupla hélice.
Uma propriedade importante do DNA é que ele pode replicar-se ou fazer cópias de si mesmo. Cada filamento de DNA na dupla hélice pode servir como modelo para guiar a duplicação da sequência de bases.
Uma análise detalhada de como funciona o PCR na detecção de deteriorantes de cerveja
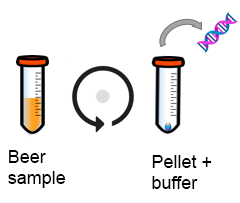
1. Separando o DNA de bactérias ou leveduras
A amostra é centrifugada para concentrar as células de deteriorantes antes de se adicionar um tampão para quebrar a parede celular
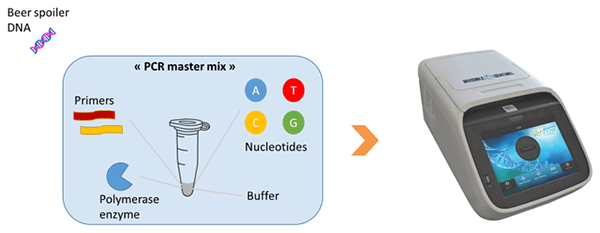
2. O DNA do deteriorante é então adicionado à "solução master mix" de PCR que contém todos os elementos necessários para a reação (replicação do DNA) e então é colocado num termociclador.
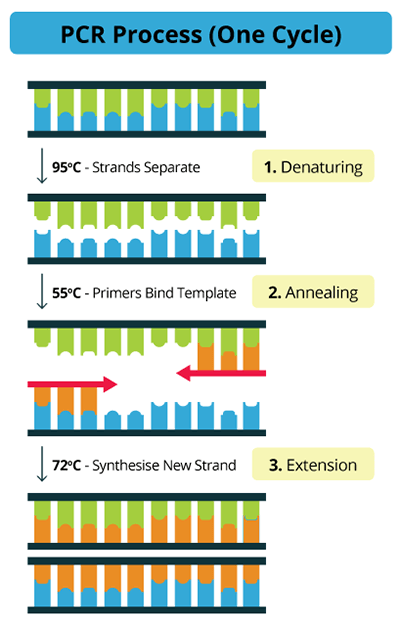 3. É aplicada uma alta temperatura para separar os 2 filamentos de DNA (Desnaturação) e, a seguir, a redução da temperatura permite que os "adaptadores específicos para o deteriorante" (primers) procurem o código de DNA correspondente e alinhem-se em frente a eles (Anelamento).
3. É aplicada uma alta temperatura para separar os 2 filamentos de DNA (Desnaturação) e, a seguir, a redução da temperatura permite que os "adaptadores específicos para o deteriorante" (primers) procurem o código de DNA correspondente e alinhem-se em frente a eles (Anelamento).
4. Quando os primers estão em posição, a enzima (Polimerase) sabe por onde começar e terminar a síntese de uma nova fita para criar uma nova dupla fita (Extensão).
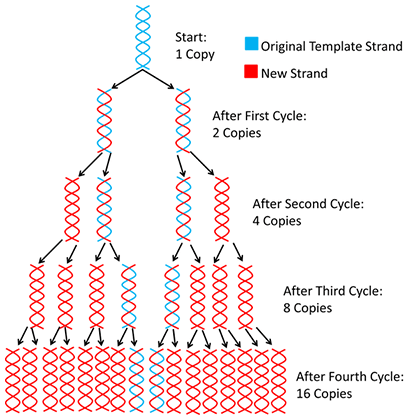
E assim, 1 cadeia passa a ser 2 após um ciclo completo. Os ciclos são então repetidos no "Termociclador"
No final de cada ciclo de PCR, o produto PCR ou amplicon aumentará exponencialmente porque as sequências de DNA recém sintetizadas podem ser utilizadas como modelo (somando-se ao modelo original de DNA). Normalmente, 20 a 30 ciclos de PCR são suficientes para alcançar um aumento de 106 a 109 dos fragmentos de ADN.
5. A leitura dos amplicons sintetizados por PCR pode acontecer de 2 modos!
a) A PCR quantitativa em tempo real ( qRT-PCR) baseia-se no princípio de que quantidades iniciais mais altas ou mais baixas de uma determinada sequência de DNA (presença de deteriorante de cerveja) levarão a concentrações mais altas ou mais baixas de amplicons, respectivamente. À medida que os amplicons são sintetizados, são incorporam um marcador por fluorescência que pode depois ser "lido" por um leitor a laser ou óptico e interpretado de acordo com o softwares que inferem a concentração dos amplicons.
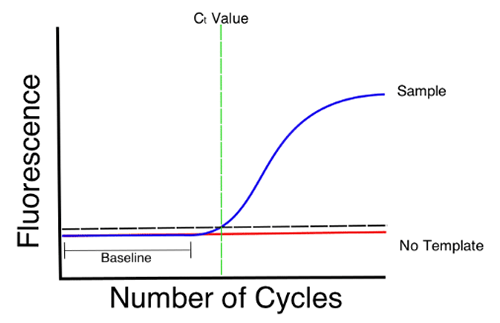
O resultado é frequentemente expresso como um valor Ct (ciclo limite) que é o número do ciclo de PCR no qual a curva da reação de uma amostra cruza um limiar previamente estabelecido. Este valor indica quantos ciclos foram necessários para detectar um sinal genuíno das suas amostras. A interpretação dos resultados pode muitas vezes ser difícil e requerer experiência para dizer com segurança se existe ou não um risco de presença de um deteriorante.
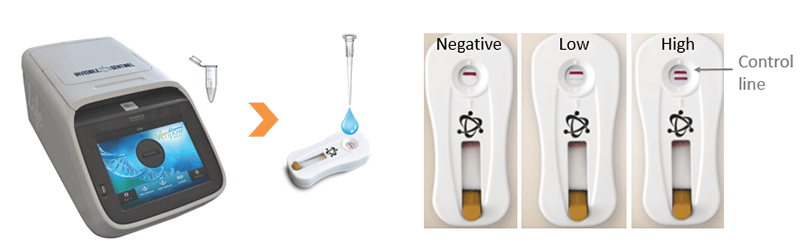
b) O PCR em endpoint (ponto final) a leitura de teste rápido baseia-se, de forma similar ao qRT-PCR, no princípio de que quantidades iniciais mais altas ou mais baixas de uma sequência específica de DNA (presença de deteriorante) conduzirão a concentrações mais altas ou mais baixas de amplicons, respectivamente. A diferença é que se realiza um número fixo de ciclos e quando cada amplicon é sintetizado, em vez de ser etiquetado com um marcador por fluorescência, liga-se a ele um biomarcador que será depois detectado por um "anticorpo" específico, presente em um teste rápido. Assim como com um "teste de gravidez", o resultado não é apenas visível com presença/ausência, mas quando presente, a intensidade da linha pode ser medida em comparação com um quadro "padrão" para determinar o valor semiquantitativo de células de deteriorantes de cerveja originalmente presentes.
A pasteurização da cerveja e subsequente controle da presença de deteriorantes por PCR
Embora, como mencionado acima, a tecnologia PCR seja rápida, sensível e específica, tornando-a uma ferramenta ideal para o laboratório da cervejaria, não é capaz de diferenciar o DNA proveniente de células vivas daquele de células mortas. Para o PCR, DNA é simplesmente DNA! Para muitas cervejarias artesanais isto não é um problema, contudo para aquelas que pasteurizam a sua cerveja, a utilização de PCR após a pasteurização (isto é, antes do engarrafamento ou do produto final) pode deixar os gestores de CQ um tanto perplexos quando obtêm um resultado positivo numa amostra.
Ele é proveniente de células vivas que sobreviveram à pasteurização e, portanto, impõem um risco para a qualidade do produto acabado ou é simplesmente o DNA remanescente após a célula ter sido morta e, embora alerte o gestor de CQ para um problema anterior, ele já não representa uma ameaça para a qualidade da cerveja durante o seu prazo de validade?
Em alguns casos, o fornecedor do PCR irá propor uma etapa de enriquecimento "pós-pasteurização" de pelo menos 24 horas antes da análise para que, caso o DNA das células mortas estiver presente, seja diluído durante o enriquecimento e obviamente não aumente durante o enriquecimento. O resultado do PCR após o enriquecimento depende, no entanto, do nível de contaminação antes da pasteurização e do tempo que o DNA liberado permanece intacto, o que dependerá de muitas variáveis diferentes da cerveja (por exemplo, pH, temperatura ...). Os chamados "falsos" positivos ainda são possíveis.
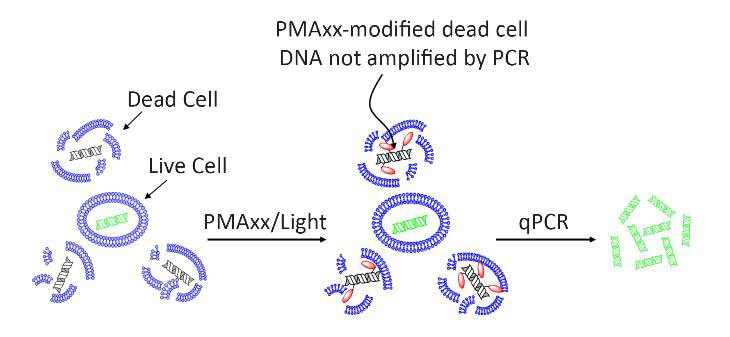
Outra abordagem é expor o DNA a uma molécula (por exemplo, PMAxx TM - Biotium, Inc.) que tem uma grande afinidade ao ácido nucleico e se liga a ele, mas que é incapaz de atravessar uma célula viável para acessar seu DNA antes da amplificação. Uma vez ligado e exposto a luz de um determinado comprimento de onda , o DNA "ligado" não pode ser amplificado no PCR. O resultado final é que, se for observado um resultado positivo, sabe-se que provém apenas de células vivas e viáveis!
Pontos-chave
• A tecnologia de Reação em Cadeia da Polimerase (PCR), é muito específica e sensível devido à virtude de detectar sequências específicas de DNA para os deteriorantes de cerveja
• Nos últimos 5 anos, o PCR foi democratizada por meio da simplificação de protocolos e da redução dos custos de investimento para o sistema.
• Atualmente, o PCR é considerado uma verdadeira ferramenta "para a produção", devido à sua capacidade de fornecer resultados no mesmo dia.
• Existem 2 abordagens à tecnologia PCR - (1) PCR endpoint (de fase final): amplificação do segmento de DNA selecionado para um número fixo de ciclos e análise subsequente do resultado. (2) qPCR: durante o processo de amplificação, análise em Tempo Real de DNA presente ciclo após ciclo.
• PCR permite tanto resultados qualitativos como quantitativos, dependendo da plataforma
• O CQ de cervejas pasteurizadas utilizando PCR requer um passo adicional para evitar a possibilidade de "falsos positivos"